Manual Peak Integration under GMP - Just a Single Aspect
During training courses, especially when customers have used other CDS systems than Empower before, I’m regulary confronted with the statement that manual peak integration, as it is e.g. technically implemented in the Empower CDS, is not acceptable from a regulatory perspective.
participants argue that if only the peak baselines and perpendiculars a drawn manually in the chromatogram, the integration cannot be clearly described and thus repeated in a reproducible way.
This was once requested during an audit or the customers was educated in this way in the past, and for this reason alone, manual peak integration has even been entirely prohibited in Empower in some laboratories.
Well, this cannot be true, as I know many Empower customers performing manual peak integration in this way and they all do pass their audits. Is the statement mentioned above therefore completely wrong?
I am regularly surprised by this statement, since nobody can explain, where this requirement is defined and what sense it has in systems like Empower.
To my knowledge, the only explicit requirement for manual peak integration is described in the ‘ICH Guideline M10 on Bioanalytica Method Validation’: Section 3.3.6: “…Original and reintegrated chromatograms and initial and repeat integration results should be kept for future reference…” In other words, all integration results once generated for a chromatogram, regardless of whether it has been generated by an integration program or manually, needs to be stored permanently and available at any time.
But even without explicit mention of manual peak integration, the general audit trail rules apply in this case, according to which the entire history for the generation of the final result must be traceable.
This core requirement is met very well in the Empower CDS and it doesn’t matter how the manual integration was implemented from a purely technical point of view, you don’t have to be able to describe it using events, because each result version is available as an independent database entry with the resulting peak integration permanently and unchangeably. A comparison of the different result versions can easily be provided visually in an overlay plot.
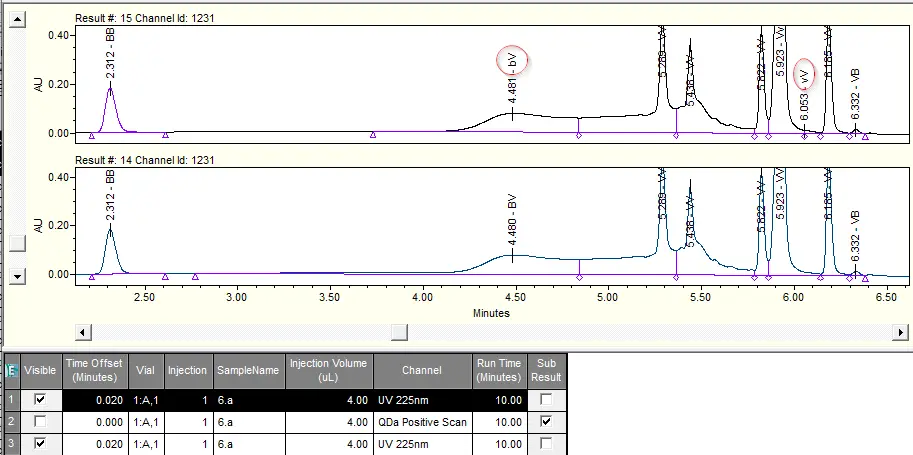
For further clarification, however, it helps to look beyond the end of one’s nose. Because there are CDS systems in which not every version of an integrated chromatogram is directly available and different versions can therefore be visually compared with one another. Only the course of changes is recorded in the history.
In such a case, of course, it must be possible to describe the peak integration in a way that the resulting integrated chromatogram can be called up from the raw data channel at any time and reproduced precisely.
In this context, the requirement described at the beginning is crucial. In such a case, for example, a modified version of the original integration method can be assigned to the re-integrated chromatogram.
One can therefore speak of a requirement that derives from the actual core requirement, as described in ICH M10, but ultimately only relates to the technical capabilities of the underlying software system. Since this is only a derived requirement, it cannot be directly transferred to a system like Empower, where the core requirement is already directly met by a different technical means.
Please note, this article does not intend to fully describe all conditions under how manual peak integration is defined and when it is allowed in a regulated environment. It’s only about the mentioned single aspect, where I wasn’t able to find anything elsewhere.
For capturing the entire topic I can recommend the article “Can We Continue to Draw the Line, from H. Longden and R.D. McDowall in LCGC Europe, December 2019, volume 32 No 12”
Ingo Green




